Myths about teaching can hold you back
- Year 10
- Edexcel
- Higher
Using models to explain state changes
I can explain why there is a difference in melting/boiling points between simple and giant covalent structures, and use models to show the shapes and structures of covalent substances.
- Year 10
- Edexcel
- Higher
Using models to explain state changes
I can explain why there is a difference in melting/boiling points between simple and giant covalent structures, and use models to show the shapes and structures of covalent substances.
Lesson details
Key learning points
- Molecular modelling represents the bonding and structure of substances.
- The way giant covalent structures melt/boil is very different to simple molecules.
- Diamond is very hard, has a very high melting point and does not conduct electricity, every atom is bonded to another.
- In graphite each atom is bonded to three others to form layers of hexagonal rings. Each atom contributes a free electron
- Simple molecules are held together by intermolecular forces, no chemical bonds hold them together.
Keywords
Simple molecular - Simple molecular substances are made up of two or more atoms covalently bonded together to form a distinct unit.
Model - Scientists use models to represent parts of the natural world that are too difficult to observe or explain directly.
Intermolecular force - Intermolecular forces are weak forces of attraction between molecules and molecular substances.
Giant covalent - A giant covalent substance has a large regular arrangement of atoms all joined together by covalent bonds.
Common misconception
Covalent bonds break in simple molecules for substances to melt/boil, because this happens in giant covalent structures.
Highlight that bonds require large amounts of energy to break, hence the very high melting points of diamond and graphite. As all atoms are bonded these must be broken to melt/boil.
To help you plan your year 10 chemistry lesson on: Using models to explain state changes, download all teaching resources for free and adapt to suit your pupils' needs...
To help you plan your year 10 chemistry lesson on: Using models to explain state changes, download all teaching resources for free and adapt to suit your pupils' needs.
The starter quiz will activate and check your pupils' prior knowledge, with versions available both with and without answers in PDF format.
We use learning cycles to break down learning into key concepts or ideas linked to the learning outcome. Each learning cycle features explanations with checks for understanding and practice tasks with feedback. All of this is found in our slide decks, ready for you to download and edit. The practice tasks are also available as printable worksheets and some lessons have additional materials with extra material you might need for teaching the lesson.
The assessment exit quiz will test your pupils' understanding of the key learning points.
Our video is a tool for planning, showing how other teachers might teach the lesson, offering helpful tips, modelled explanations and inspiration for your own delivery in the classroom. Plus, you can set it as homework or revision for pupils and keep their learning on track by sharing an online pupil version of this lesson.
Explore more key stage 4 chemistry lessons from the Chemistry of carbon unit, dive into the full secondary chemistry curriculum, or learn more about lesson planning.

Equipment
Molecular modelling kits.
Licence
Lesson video
Loading...
Prior knowledge starter quiz
6 Questions
Q1.Below, there are six different models for representing the bonding and structure in the compound ethane. Match the image to the correct type of model.
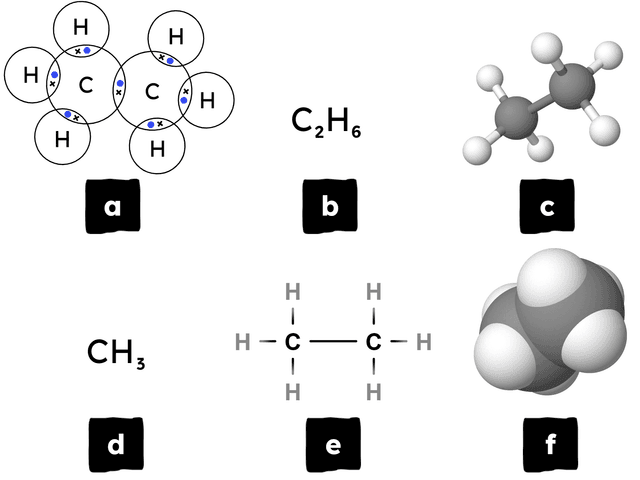
dot-and-cross diagram
molecular formula
ball-and-stick model
empirical formula
displayed formula
space-filling model
Q2.What do the dots and crosses typically represent in dot-and-cross diagrams?
Q3.Which of the following are limitations of the empirical formula when representing molecules?
Q4.Which model would not be appropriate to represent the bonding and/or structure in metals?
Q5.What advantage does the ball-and-stick model have over the space-filling model?
Q6.Which representation is most suitable for illustrating the arrangement of ions in a giant ionic lattice?
Assessment exit quiz
6 Questions
Q1.Which properties of diamond are explained by the fact that each carbon atom is strongly bonded to four other carbon atoms in a rigid structure?
Q2.What type of forces hold simple molecules together?
Q3.One model of graphite shows it having free moving, delocalised electrons. This means graphite is an electrical ?
Q4.Match each substance to the correct statement about it's structure and/or properties.
doesn't conduct electricity as every atom is strongly bonded to others
hexagonal rings of carbon atoms, arranged in layers
made of simple molecules with low melting and boiling points
a mixture of carbon-containing molecules
Q5.Match the key words to their definitions.
distinct units made up of two or more atoms covalently bonded
represent parts of the natural world that are too difficult to observe
weak forces of attraction between molecules
large regular arrangement of atoms all joined by covalent bonds
