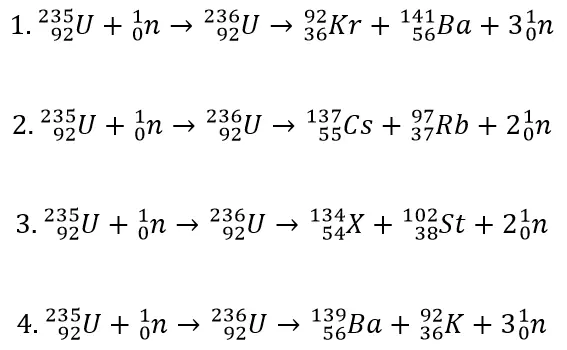Myths about teaching can hold you back
- Year 11
- AQA
- Foundation
Nuclear fission and fusion
I can explain how energy can be transferred from the nuclei of atoms to cause heating.
- Year 11
- AQA
- Foundation
Nuclear fission and fusion
I can explain how energy can be transferred from the nuclei of atoms to cause heating.
Lesson details
Key learning points
- Nuclear fission is the splitting of a large and unstable nucleus such as uranium or plutonium.
- A neutron fired at a uranium nucleus can cause nuclear fission that splits the nucleus and emits 2 or 3 more neutrons.
- The motion of all the particles shot out from a nuclear fission can cause heating by making other particles move faster.
- Neutrons shot out from a nuclear fission can split more uranium nuclei in a chain reaction.
- Nuclear fusion is the joining of two light nuclei to form a heavier nucleus with the creation of gamma radiation.
Keywords
Nuclear fission - the splitting of large nuclei to form pairs of smaller nuclei and some free neutrons, with the release of energy
Daughter nuclei - the smaller nuclei produced during nuclear fission
Chain reaction - a series of nuclear fission events, with each one triggered by a neutron from a previous nuclear fission
Nuclear fusion - the combining of small nuclei at very high temperatures with the release of energy
Common misconception
Radioactive isotopes disappear when they decay, so that in one half-life half the mass is lost.
Emphasise that the mass of the nuclei at the end of a nuclear fission or fusion are almost identical to the masses of those at the start. There is a very small difference, with mass being converted to energy according to E = mc².
To help you plan your year 11 physics lesson on: Nuclear fission and fusion, download all teaching resources for free and adapt to suit your pupils' needs...
To help you plan your year 11 physics lesson on: Nuclear fission and fusion, download all teaching resources for free and adapt to suit your pupils' needs.
The starter quiz will activate and check your pupils' prior knowledge, with versions available both with and without answers in PDF format.
We use learning cycles to break down learning into key concepts or ideas linked to the learning outcome. Each learning cycle features explanations with checks for understanding and practice tasks with feedback. All of this is found in our slide decks, ready for you to download and edit. The practice tasks are also available as printable worksheets and some lessons have additional materials with extra material you might need for teaching the lesson.
The assessment exit quiz will test your pupils' understanding of the key learning points.
Our video is a tool for planning, showing how other teachers might teach the lesson, offering helpful tips, modelled explanations and inspiration for your own delivery in the classroom. Plus, you can set it as homework or revision for pupils and keep their learning on track by sharing an online pupil version of this lesson.
Explore more key stage 4 physics lessons from the Nuclear physics unit, dive into the full secondary physics curriculum, or learn more about lesson planning.

Equipment
None required.
Licence
Lesson video
Loading...
Prior knowledge starter quiz
6 Questions
Q1.Which of the following particles are found in the nucleus of an atom?
Q2.An isotope can be defined as …
Q3.What are the possible differences between substances that contain isotopes of the same element?
Q4.Which particle affects the stability of a nucleus, and what factors determine if a nucleus is stable or unstable?
Q5.Which of these can be released during nuclear reactions such as nuclear decay?
Q6.Which of the following statements accurately describes the changes that occur in the nucleus during radioactive decay?
Assessment exit quiz
6 Questions
Q1.What is nuclear fission?
Q2.Which of the following nuclear equations could represent the fission of a uranium–235 nucleus?