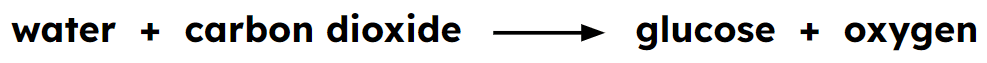Myths about teaching can hold you back
- Year 8
Characteristics of chemical reactions
I can describe some of the changes that indicate a chemical reaction is taking place.
- Year 8
Characteristics of chemical reactions
I can describe some of the changes that indicate a chemical reaction is taking place.
Lesson details
Key learning points
- Characteristics of chemical reactions include changes in colour, bubbling, or a change of temperature.
- Bubbles appear when a gas is produced in a chemical reaction.
- In a chemical reaction, atoms rearrange themselves to form new substances.
- Generally, chemical reactions are irreversible.
Keywords
Chemical reaction - when atoms are rearranged to make new products and changes can be observed e.g. bubbles of gas forming.
Irreversible reaction - occurs in one direction only where the reactants are converted into the products.
Reactants - elements or compounds before a reaction takes place. Reactants are found on the left hand side of a chemical equation.
Products - elements or compounds that are formed after a reaction has taken place. Products are found on the right hand side of a chemical equation.
Common misconception
Pupils do not understand chemical reactions as changes, they considered them to be ‘events’. They focus on observable features e.g. colour change rather than formation of a new substance.
Use questioning to encourage pupils to make links between their observations as evidence that a new substance is formed when substances undergo chemical reactions.
To help you plan your year 8 science lesson on: Characteristics of chemical reactions, download all teaching resources for free and adapt to suit your pupils' needs...
To help you plan your year 8 science lesson on: Characteristics of chemical reactions, download all teaching resources for free and adapt to suit your pupils' needs.
The starter quiz will activate and check your pupils' prior knowledge, with versions available both with and without answers in PDF format.
We use learning cycles to break down learning into key concepts or ideas linked to the learning outcome. Each learning cycle features explanations with checks for understanding and practice tasks with feedback. All of this is found in our slide decks, ready for you to download and edit. The practice tasks are also available as printable worksheets and some lessons have additional materials with extra material you might need for teaching the lesson.
The assessment exit quiz will test your pupils' understanding of the key learning points.
Our video is a tool for planning, showing how other teachers might teach the lesson, offering helpful tips, modelled explanations and inspiration for your own delivery in the classroom. Plus, you can set it as homework or revision for pupils and keep their learning on track by sharing an online pupil version of this lesson.
Explore more key stage 3 science lessons from the Understanding chemical reactions unit, dive into the full secondary science curriculum, or learn more about lesson planning.

Equipment
See additional materials for equipment list
Content guidance
- Risk assessment required - equipment
Supervision
Adult supervision required
Licence
Lesson video
Loading...
Prior knowledge starter quiz
6 Questions
Q1.When a substance changes from a solid to a liquid, it is called .
Q2.Which of the following is an example of a chemical change?
Q3.A change is one that does not produce new substances and can often be reversed.
Q4.Which of the following represents a physical change?
Q5.What is the main difference between a physical change and a chemical change?
Q6.An is a substance made of one type of atom.
Assessment exit quiz
6 Questions
Q1.What is a chemical reaction?
Q2.Where are products found in a chemical equation?